When it comes to medical devices, there’s absolutely zero room for error. You’re dealing with human lives, not just products on a shelf. One faulty design or overlooked issue could mean chronic pain, permanent injury, or worse.
Yet, time and again, we hear stories about devices malfunctioning, getting recalled, or causing unexpected complications. This year, so far, the US FDA has already recalled 50 medical devices. Early alerts have been raised against six other such devices, meaning that these medical products pose a high risk when being removed or corrected. Moreover, research shows that the number of medical device recalls has increased over the years.
The truth is, many of these problems in medical devices could have been prevented. It’s not just about designing smart gadgets that work. It’s about designing them with care, testing them with rigor, and listening to the people who use them.
If you’re a medical device manufacturer, you have a responsibility that stretches far beyond the lab. People trust you with their bodies, their health, and sometimes their futures.
So, if you’re looking to improve your product’s safety and quality, it starts with a mindset. Not a checklist, not a regulatory document, but the willingness to dig deeper and ask tough questions.
Let’s break down the measures you can take to enhance the overall safety and quality of your medical devices.
Design with the End User in Mind
The first mistake many companies make is designing for approval, not for people. Regulatory clearance becomes the goal instead of patient well-being. Sure, your device might tick every box required by the FDA or other oversight bodies. But what happens when it hits the real world?
Doctors don’t always use tools the same way engineers imagined. Patients vary. Environments vary. Situations aren’t clean and controlled. This is where so many products fall apart.
Instead of designing from a lab bench, start from the hospital floor or the patient’s home. Talk to nurses. Watch procedures. Study what makes some devices easier to use and others more prone to errors.
Engineers love function. But safety often lives in the small details like grip texture, labeling clarity, durability, and even noise levels. If you build a product thinking only about how it should work, you’re already a few steps behind. Build it for how people will actually use it.
Communicate Across Departments
One of the quiet killers of medical device quality is siloed communication.
Engineering doesn’t talk to marketing. Manufacturing doesn’t talk to design. Customer feedback doesn’t reach product development. And when something does go wrong, no one really knows whose job it is to fix it.
That’s a massive problem.
To avoid this, you need transparency from top to bottom. Not just updates and reports, but real conversations.
Your design team should be part of your post-market surveillance process. Your engineers should know about customer complaints. Everyone involved in the device lifecycle should be hearing the same story from the same people.
Problems fester when they’re only half-seen. If your departments don’t feel connected, your product will eventually reflect that disconnect.
Do Rigorous Quality Checks and Don’t Cut Corners
Now let’s talk about quality checks, not the kind you breeze through just to meet a deadline. We’re talking about deep, no-compromise inspections.
Before a device gets anywhere near a patient, it should be tested under conditions far tougher than it will ever face in real life. We’re talking about stress tests, fatigue simulations, extreme temperature trials, and so on. If a component is going to fail, it’s better to catch it in your lab than in someone’s body.
But here’s where things often fall apart. Under pressure to release a product quickly, some teams reduce testing scope or fast-track results. The idea is that real-world feedback will fill in the gaps.
However, real-world feedback can come at the cost of someone’s health. A rushed test is never worth the risk. The better option? Build time into your development cycle for failure. You need space to test, tweak, and test again. Quality should never feel like an afterthought or a luxury.
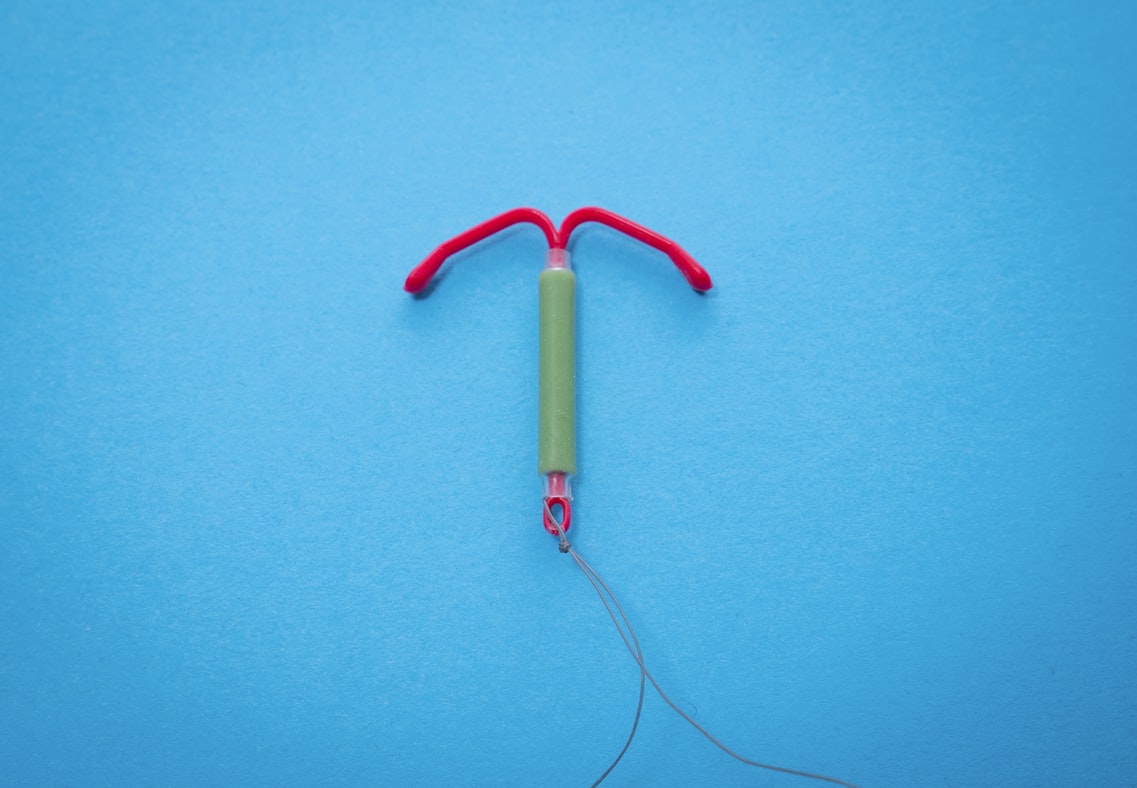
The Paragard IUD is a hard lesson in what happens when quality checks miss the mark. On paper, this intrauterine device seemed like a win. It’s a non-hormonal birth control that lasts for years. But what started as a promising product quickly spiraled into a legal and medical nightmare.
Reports began surfacing about the Paragard IUD breaking during removal. For many women, that meant surgery, infection, infertility, or lasting pain. These Paragard IUD complications weren’t rare outliers. They reflected a deeper problem in the product’s design or materials. And the lawsuits followed.
The number of Paragard IUD lawsuits has climbed sharply, as women seek answers and accountability. Paragard lawyers are now helping victims navigate what has become a complicated and emotional legal process. Some are even discussing Paragard IUD lawsuit settlement amounts that reflect the serious harm done.
Had there been more thorough stress testing on device removal scenarios, some of these issues could have been prevented. The fallout from the Paragard IUD shows just how important real, rigorous quality checks are. These checks are vital not just for your reputation, but for the health and safety of the people you serve.
Invest in Ongoing Training for Technicians and Staff
Your devices might be engineered with surgical precision. However, if your staff doesn’t know how to inspect them properly, something will still go wrong.
Quality isn’t a static target. It requires eyes that are trained, hands that are steady, and people who actually care about catching flaws. Many companies assume that once a team member knows the basics, that’s enough. It’s not.
Devices evolve. Materials change. And so do regulatory expectations. Your team needs updated training, just like your software needs updates. This isn’t about micromanaging every worker. It’s about giving people the tools to spot issues before they become disasters.
A well-trained technician can mean the difference between a bad batch of products slipping through and a critical catch that saves someone’s life. That includes your leadership.
Managers and executives need to understand quality just as much as the people on the floor. They set the tone. If your leadership sees inspections as roadblocks, your team will too. But if you build a culture where quality is part of everyone’s job, it becomes easier to maintain high standards.
Keep Listening After the Product Launch
So you’ve launched the product. The press releases or ads are out. The packaging looks great. Your sales team is buzzing. But this is not the finish line. In fact, it’s where the real test begins.
Globally, the medical device design and development services market is worth $11.5 billion. Yet, problems can arise with the product’s design and functionality, so each device type needs its own post-launch oversight.
What happens after your product is out in the world is what really tells you how safe and effective it is. You can’t afford to turn your ears off at this stage.
Set up systems to collect feedback in real time. Encourage doctors, nurses, and patients to report issues. And don’t treat complaints as annoyances. They’re early warnings. The faster you act on them, the fewer people get hurt. Yes, you’ll need a post-market surveillance system, not just because regulations say so, but because it’s your ethical duty.
Also, be honest. If a product is showing patterns of failure, acknowledge it publicly. Hiding flaws only backfires, as was seen previously with the Paragard IUD case. Transparency may sting in the short term, but it builds trust over time. And in healthcare, trust is everything.
Collaborate with Third-Party Experts
Sometimes, being too close to your product can make you blind to its problems. That’s where outside eyes help.
Independent auditors, medical advisors, and safety consultants are the people who can offer you a fresh perspective. Don’t wait until a lawsuit or recall forces you to bring them in. Use them early and often.
External reviews can reveal weak spots in your process that you’ve stopped noticing. Maybe your documentation is sloppy. Maybe your packaging creates usability issues. Maybe your device looks great under a microscope, but fails in high humidity. You don’t know what you don’t know until someone else helps you see it.
The best manufacturers are humble enough to invite critique. They don’t get defensive. They get better.
Focus on Ethical Responsibility, Not Just Compliance
Many companies see compliance as the finish line. If it passes FDA checks, it’s good to go, right? Not quite. Compliance is the bare minimum. Ethics go beyond that. Just because you can legally market a product doesn’t mean you should.
Ask yourself: Would you feel safe using this device on your child? Your partner? Yourself? If the answer is anything short of a strong yes, it’s time to rethink your standards.
Ethics in medical manufacturing isn’t just about avoiding fines. It’s about taking personal responsibility for what you create. People’s lives are in your hands. Your decisions today can echo for years. That’s a heavy responsibility, one worth taking seriously.
Frequently Asked Questions (FAQs)
Why are medical devices hard to manufacture?
Medical devices are difficult to manufacture due to strict regulatory requirements, the need for precise functionality, and complex materials. Each device must meet safety standards set by organizations like the FDA or ISO. Small design errors can lead to serious health consequences. Manufacturers must balance innovation with reliability, making the development process both time-consuming and costly.
What makes quality checks so vital when designing and manufacturing medical devices?
Quality checks are essential because even minor defects can endanger patients’ lives. These checks ensure devices perform as intended under real-world conditions. They help identify design flaws early, reducing the risk of recalls and lawsuits. Without rigorous quality control, devices may fail during use, leading to serious injuries or death.
How are safety tests conducted for medical devices?
Safety tests for medical devices often involve simulated use, mechanical stress testing, and biocompatibility assessments. Clinical trials may also be required to observe performance in actual patients. Regulatory bodies demand documentation and data from these tests to approve the device. This process ensures the device is safe, effective, and poses minimal risks.
Are healthcare professionals involved in the design and development of medical devices?
Yes, healthcare professionals often collaborate in designing and developing medical devices. Their insights help ensure the product is practical, user-friendly, and meets clinical needs. They can identify gaps or inefficiencies in current devices that designers may overlook. Their involvement also improves safety and functionality from a real-world usage perspective.
What kind of medical devices pose the most risks?
Implantable devices like pacemakers, spinal stimulators, and intrauterine devices pose the most risks. These remain inside the body for long periods and can cause complications if they malfunction or are difficult to remove. Issues like infections, device migration, or breakage can have serious health consequences. Their complexity makes both design and post-market monitoring critical.
Are medical devices tested directly on humans?
Yes, medical devices are often tested on humans, but only after passing lab and animal testing. This stage is known as a clinical trial and is strictly regulated. Human testing helps evaluate the device’s safety, effectiveness, and usability in real-world medical settings. These trials usually involve volunteers and are conducted under close ethical and medical supervision.
At the end of the day, building safer medical devices isn’t about perfect materials or flawless technology. It’s about intention. The intention is to design better. To test deeper. To listen longer. To fix what’s broken without waiting for lawsuits or headlines. That mindset separates the companies that are truly making a difference from those just trying to stay afloat.
If you’re part of this industry, you already know how hard the work can be. There are deadlines, budgets, and an endless list of regulations. But above all that, there’s a real human cost when things go wrong. That’s the part you can never lose sight of.
So, don’t settle for checking boxes. Don’t wait for a scandal to push you into action. Start now. Build better. Listen more. And never forget that behind every device, there’s a person who’s counting on it to work and counting on you to get it right.