According to the definition of fossil fuels, the answer to the question “Is natural gas a fossil fuel” is “yes”.
Many years ago, when prehistoric plants and animals died, they were gradually buried under layers of rock and soil. During millions of years, the heat and pressure of Earth’s crust decomposed organisms into one of the three main classes of fuel, including oil (also called petroleum), natural gas, and coal. Because these fuels are created from the remains of dead plants and animals are called fossil fuels.
What Is Natural Gas?
Natural gas is a source of fossil energy that has formed deep underground. Natural gas contains various compounds. The main constituent of natural gas is methane, the chemical formula CH₄, a chemical compound including one carbon atom and four hydrogen atoms. Natural gas also contains fewer amounts of natural gas liquids (NGL, hydrocarbon gas liquids) and non-hydrocarbon gases, like carbon dioxide and water vapor. We employ natural gas as a fuel and produce materials and chemicals.
How Is Natural Gas Formed?
Hundreds of millions to millions of years ago and over long periods, the remains of animals and plants (such as diatoms) formed in thick layers on the earth’s surface and ocean floors, and in some cases, they were mixed with sand, silt, and calcium carbonate. Gradually, these layers were buried beneath sand, silt, and rock. Pressure and heat turned some carbon and hydrogen-rich material into oil (petroleum), some into coal, and some into natural gas.
Types of Natural Gas Based on Where It Is Found
In some areas, natural gas is moved inside huge cracks and spaces between overlying rock layers. The natural gas in this type of structure is sometimes called conventional natural gas. In other places, natural gas takes place in the small pores (spaces) within some forms of shale, sandstone, and other sedimentary rock types. This natural gas type is referred to as shale gas or tight gas and is sometimes called unconventional natural gas.
Natural gas formation also occurs with crude oil deposits, and this type of natural gas is called associated natural gas. Natural gas deposits are located on land, and some of them are offshore and deep beneath the ocean floor. The type of natural gas discovered in coal deposits is known as coal bed methane.
How to Find Natural Gas?
The search for natural gas starts with geologists studying the structure and processes of the earth. They determine the types of geologic formations that are probable to hold natural gas deposits. Geologists usually use seismic surveys on land and in the ocean to find suitable locations to drill natural gas and oil wells. Seismic surveys generate and measure seismic waves to obtain information about the geology of rock formations.
Seismic surveys on land may apply a thumper truck with a pad that vibrates the ground to produce seismic waves in the underlying rock. Sometimes little amounts of explosives are utilized. Seismic surveys conducted in the ocean use sound explosions that generate sonic waves to explore the geology beneath the ocean floor.
If the outputs of seismic surveys show that a site has the potential to produce natural gas, an exploratory well will be drilled and tested. The test results provide information on the quality and quantity of natural gas accessible in the resource.
Drilling Wells of Natural Gas
If the outcomes of a test
confirm that a geologic formation has enough natural gas for production and profitability, and one or more production or development wells will be drilled. Wells of natural gas may be drilled vertically and horizontally into natural gas-bearing structures. In conventional natural gas deposits, the natural gas usually flows easily up to the surface through wells.
In some countries, natural gas is generated from shale and other types of sedimentary rock formations by pushing water, chemicals, and sand into a well under high pressure. This process is called hydraulic fracturing or fracking and is sometimes known as unconventional production. It breaks the formation, releases natural gas from the rock, and makes the natural gas flow upward from the wells to the surface. Upside the well on the surface, natural gas is collected in pipelines and delivered to natural gas processing plants.
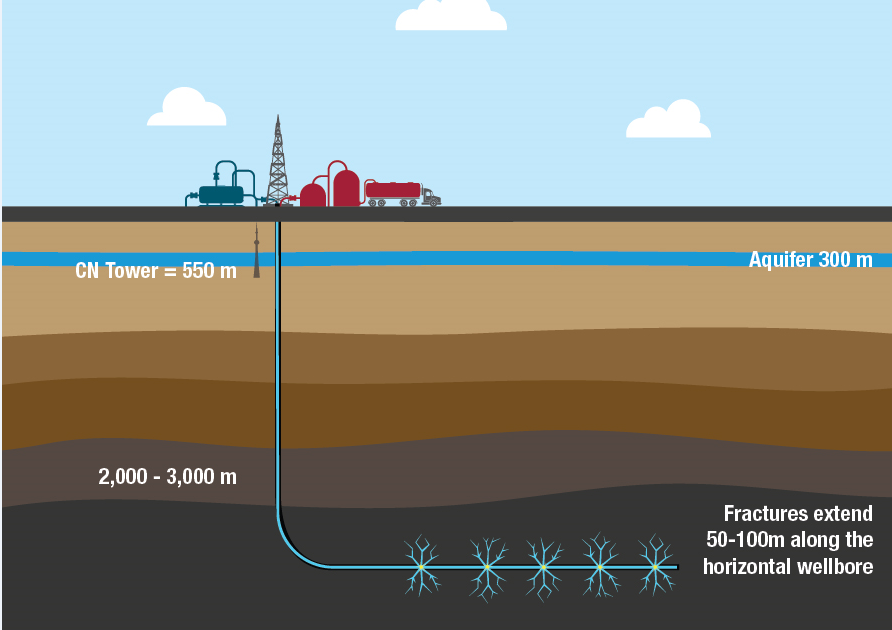
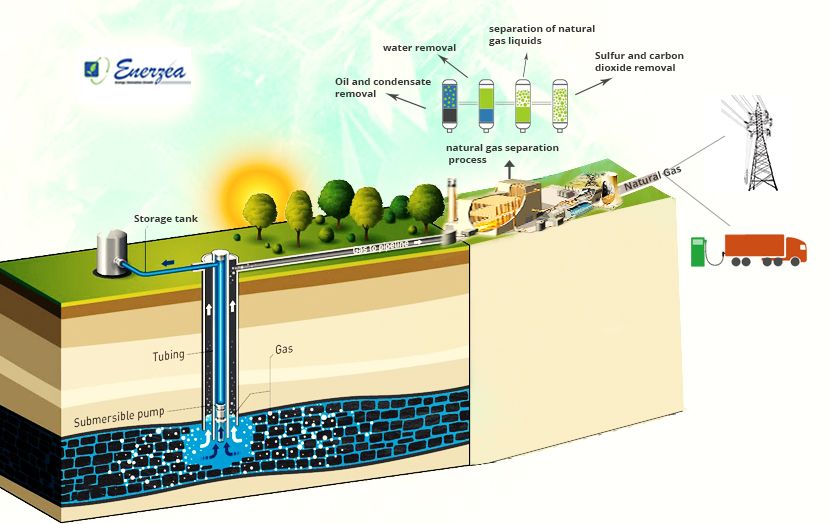
The Process Done on Natural Gas to Be Consumed
Natural gas is widely used for domestic, and industrial heating, electricity generation, and industrial chemical processes. The path that is taken from rocks underground to the homes, offices, or schools is full of possible problems. The extracted natural gas cannot reach the consumer directly without the necessary measures. Therefore, steps must be taken to be able to be transmitted to the user.
Separation of Excess Compounds
Natural gas extracted from natural gas or crude oil wells is known as wet natural gas because, along with methane, it usually includes natural gas liquids (NGL) and water vapor. Wellhead natural gas may also contain non-hydrocarbon substances, including sulfur, nitrogen, helium, hydrogen sulfide, and carbon dioxide, most of which have to be eliminated from natural gas composition before it is sold to consumers.
Natural Gas Liquids
Natural gas liquids (NGL) are natural gas components that are separated from the gas in the form of liquids. This separation happens in a field facility or a gas processing plant by condensation, absorption, or other methods. There are different types of natural gas liquids and many various applications for NGL products like heating, cooking, fuels, and plastics. A problem with NGLs is that they can be costly to store, handle, and transport.
Natural gas liquids as separate products are valuable, and the removal of NGL from natural gas is profitable. The liquids are first withdrawn from the natural gas and then separated into various components. Natural gas liquids belong to the family of hydrocarbons. A hydrocarbon is a molecule comprised entirely of carbon and hydrogen.
NGLs are more broadly available with progress in drilling techniques. NGLs are employed in petrochemical feedstock that is converted into different chemical-based products. However, as said before, NGLs are expensive to control, store, and transport requiring specific equipment, storage, and trucks. NGLs need high pressure or low temperature to keep their liquid state for shipment. Increased use of NGL has led to more demand for processing plants separating NGLs from natural gas.
NGLs include Ethane, propane, butane, isobutane, and pentanes, each of which is used in some applications covering almost all economic sectors. NGLs are utilized as inputs for petrochemical plants, fired for heating and cooking, and mixed into vehicle fuel. Higher crude oil prices have pushed up the price of NGL, which in turn has provided an incentive to drill for liquids-rich resources with considerable NGL content.
Transportation from Wellhead to Consumers
From the wellhead, natural gas is transferred to processing plants where water vapor and non-hydrocarbon substances are removed, and NGL is departed from the wet gas and sold individually. Some ethane often remains in processed natural gas. The separated NGLs are known as natural gas plant liquids (NGPL). Processed natural gas is also called dry, consumer-grade, (or sometimes pipeline-quality) natural gas.
Some wellhead natural gas is dry enough to meet pipeline transportation standards without being processed. Chemicals named odorants are added to natural gas to detect leaks in natural gas pipelines. Dry natural gas is piped to underground storage fields or distribution companies and later to consumers.
In places where natural gas pipelines are not available to eliminate natural gas produced from oil wells, the natural gas can be re-injected into the oil-bearing structure, or it can be discharged or burned (flared). Re-injecting unmarketable natural gas can help to keep pressure in oil wells to enhance oil production.
Coal bed methane can be elicited from coal reserves before or during coal mining and added to natural gas pipelines without any particular treatment.
So, in short, the following steps are taken to get natural gas from our underground to our homes:
- After extraction, natural gas is usually sent via small pipelines to plants for processing.
- The different hydrocarbons and fluids are separated from the purely natural gas to create “dry natural gas.”
- This high-quality processed gas is transported through pipelines to distribution centers for use. However, sometimes it is stored in underground reserves to be used later.
Disadvantages of Natural Gas
At many stages of the described process, methane, as a potent heat-trapping greenhouse gas, can leak into the atmosphere through faulty pipes and other infrastructure. These leaks can be considerable and significantly hasten global warming. These leaks and all the heat they help trap are likely to destroy the claimed benefits.
There are two types of natural gas: dry and wet. The latter is as much related to refrigerants and plastic production as it is to the electrical outlets in your home.
The major component of dry nature is methane, but wet gas also includes compounds such as ethane and butane. These natural gas liquids are separated as described above. Then, they are used at sites like ethane cracker plants to make products like plastics.
Unfortunately, cracker plants and other components of gas infrastructure produce volatile organic compounds (VOCs) like benzene that can cause cancer in humans. These chemicals have a way of entering the air and water that local communities and workers breathe and drink every day.
Although there is a fact that an efficient natural gas power plant releases about 50 percent less carbon dioxide (CO2) when combustion occurs than a conventional coal-based power plant, 50% less CO2 isn’t zero CO2, and it is still a concern with environmentalists. Reaching net-zero greenhouse gas (GHG) emissions is essential to our long-term health of us by the second half of this century.
Also, CO2 is not the only harmful greenhouse gas emission produced by natural gas development. This brings us back to methane.
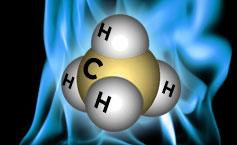
Methane is a very potent greenhouse gas. In the atmosphere, Methane is relatively short-lived compared to carbon, and only around 20 percent of the methane released today will remain in the atmosphere after 20 years. However, when it first enters the atmosphere, it is approximately 120 times stronger than CO2 in trapping heat and 86 times more potent in 20 years.
Therefore, while methane does not remain in the atmosphere as long as carbon dioxide, it is much more destructive to the climate because of its effectiveness in absorbing heat.
Buy Equipment or Ask for a Service
By using Linquip RFQ Service, you can expect to receive quotations from various suppliers across multiple industries and regions.
Click Here to Request a Quotation From Suppliers and Service Providers
Read More on Linquip
- Difference Between Renewable Energy and Fossil Fuels
- The Main Disadvantages of Natural Gas (Guide in 2023)
- Quick Guide: Advantages Of Natural Gas
- Is Natural Gas Renewable: A Clear Answer
- Important Detailed Information About The Types of Fossil Fuels
- Types of Fossil Fuels: All You Should Know
- What are the Disadvantages of Fossil Fuels?
- Advantages of Fossil Fuels
- Renewable Energy vs Fossil Fuels | How do They Compare?



